|
The device is also indicated for in-stent restenosis, or the re-narrowing of a vessel following the placement of a stent. each year, representing a growing market of over $500 million. It is designed to work on a variety of lesion types from soft plaque to severely calcified, according to the company.ĪngioDynamics estimates that more than 200,000 atherectomy procedures are performed in the U.S. 
Yoel Zabar, CEO of Eximo Medical, commented, “We believe that AngioDynamics’ existing resources and commercial acumen will enable a large number of patients to benefit from Eximo’s laser technology, and we look forward to extending its utility into other indications within vascular and elsewhere.“The market is ripe for disruption, and the level of precision, safety, and efficiency offered to physicians by this laser technology creates a substantially differentiated alternative to legacy atherectomy devices,” Clemmer said.Įximo’s 355-nm wavelength platform delivers short, high-powered laser pulses through fiber-optic cables to help remove plaque from inside the blood vessel while lowering the risk of perforating the artery. Angio adds, “This technology is a safe, fast, and effective system that clinical studies have shown to be effective in treating lesions ranging from soft plaque to severely calcified.” The technology addresses the risk of perforation through tissue selectivity, addresses the risk of embolization to the patient through the availability of aspiration and is indicated to provide treatment for In-Stent Restenosis (ISR), which is the gradual re-narrowing of the artery after a blockage has been previously treated with a stent.Īdditionally, Eximo’s laser technology can treat a variety of PAD lesion types both above- and below-the-knee. Physicians provide atherectomy treatment to over 200,000 Americans annually, and Angio says Eximo’s laser technology “represents a technological breakthrough among medical devices used in atherectomy procedures to treat PAD today.” In acquiring Eximo and its laser technology, AngioDynamics enters a growing $500-plus million market with an innovative medical solution that offers greater versatility, a wider range of treatments, and strong clinical data to support its efficacy.Įximo’s laser technology is said to be the only system capable of delivering short, high-powered pulsed-laser energy in 355nm wavelength without compromising the integrity of its fiber optic cables during atherectomy procedures. “The market is ripe for disruption, and the level of precision, safety, and efficiency offered to physicians by this laser technology creates a substantially differentiated alternative to legacy atherectomy devices.” “The acquisition of Eximo brings a remarkable, foundational technology to our portfolio that will change the way caregivers deliver treatment to patients with PAD,” commented Jim Clemmer, President and CEO of AngioDynamics. The Eximo technology complements AngioDynamics’ thrombus management and venous insufficiency technologies. This deal expands AngioDynamics’ existing vascular treatments and therapies product portfolio by adding Eximo’s proprietary laser technology, which has received 510(k) clearance for use in the treatment of Peripheral Artery Disease (PAD). The transaction is being funded exclusively through the use of cash on hand. The financial terms of the deal are $46 million in up-front consideration with up to a further $20 million of contingent consideration. 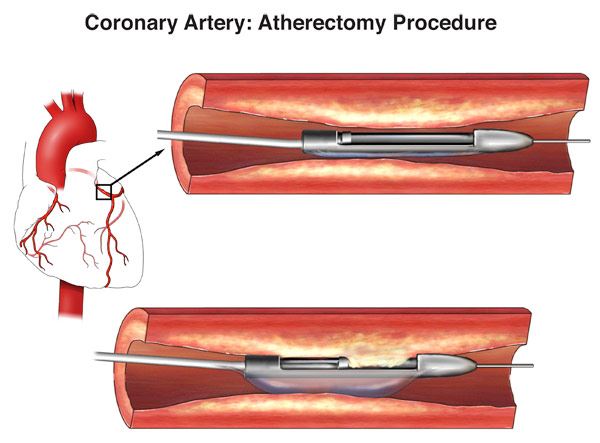
AngioDynamics, based in Latham, NY, a developer of innovative, minimally invasive medical devices for vascular diseases and oncology, has acquired Eximo Medical, a medical device company, and its proprietary 355nm wavelength laser-technology platform. Eximo’s laser tech is certified for the treatment of Peripheral Artery Disease (PAD).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
